A longstanding interest of the lab is to study mechanisms through which a protein called apolipoprotein E (ApoE) contributes to suppress the progression of atherosclerosis and promote its regression in an isoform-specific manner. To this end, we study mouse models of conditional apoE expression developed in our laboratory that we have termed HypoE mice.
Our recent studies revealed that apoE has a capacity to suppress atherosclerosis beyond its recognized ability to reduce plasma lipid levels. Cellular apoE expression in myeloid cells was found to modulate microRNA biogenesis, resulting in increased levels of microRNA-146a thereby enhancing a natural negative feedback mechanism to suppress NF-kB driven inflammation and atherosclerosis.
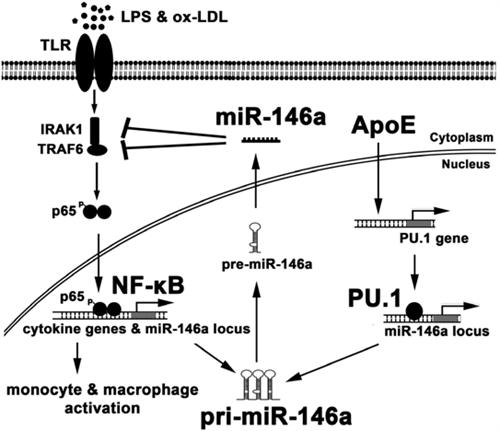
| ApoE enhances PU.1-dependent miR-146a transcription to suppress NF-κB-driven monocyte and macrophage activation and thereby inflammation and atherosclerosis |
Our findings also demonstrated that anti-atherogenic properties of apoE could in part be reproduced through the systemic delivery of microRNA-146a mimics in apoE deficient mice:
Apolipoprotein E Enhances MicroRNA-146a in Monocytes and Macrophages to Suppress Nuclear Factor-κB–Driven In ammation and Atherosclerosis
Collectively, results of our studies introduced a new level of appreciation for apoE’s capacity to serve as an anti-inflammatory molecule.
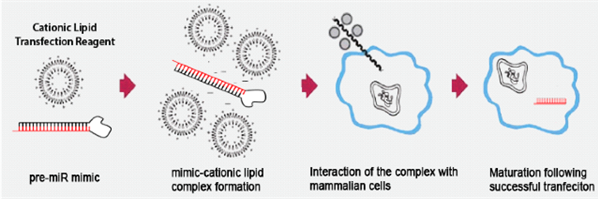
| Transfection of microRNA mimics using cationic lipid particles results in their efficient uptake and maturation into functional microRNA in cultured immune cells and monocyte/macrophages in vivo |
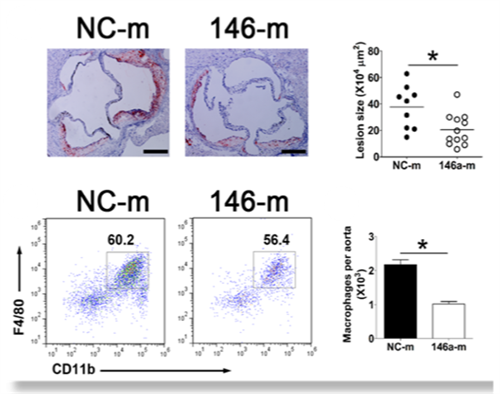
| In vivo delivery of miR-146a mimetics in lipid particles suppresses atherosclerosis in hyperlipidemic Apoe–/–mice as assessed histologically and through the enzymatic release of immune cells from the aorta followed by the detection with FACS |
Our current studies explore the influence that apoE exerts on the regulated release of microRNA into microvesicles called “exosomes” to alter their capacity to serve as extracellular mediators of inflammation and atherosclerosis. A major study underway in the laboratory explores whether infusions of myeloid-cell derived exosomes can serve to control Th1-driven inflammation and atherosclerosis in an apoE-dependent manner.
Our long-term goal is to develop new therapeutic approaches for atherosclerosis by harnessing immune-regulatory pathways controlled by apoE.